About the WCET™ International Ostomy Guideline (Third edition).
Methodology
Methodology protocol
The WCET™ IOG Panel developed and published a methodology protocol before commencing the IOG (Third edition) development. In brief, the steps involved included:
Developing the clinical questions with stakeholder input
Systematic literature searches for new evidence and incorporating evidence from the previous editions
Evaluating the evidence
Summarising the evidence using an adapted evidence-to-decision framework
Making recommendations and good practice statements
A formal consensus for good practice statements
Seeking input from enterstomal therapy (ET)/ostomy/stoma nurses and in WCET™ delegate nations, colorectal surgeons aligned with ISUCRS, individuals living with a stoma and professional ostomy organisations
Stakeholder feedback.
Developing clinical questions
The WCET™ IOG Panel undertook an international survey to ensure that the clinical questions would address stakeholder interest.
An anonymous online stakeholder survey was developed and distributed to ET/ostomy/stoma nurses and physicians in WCET™ and ISUCRS, individuals living with a stoma and professional ostomy organisations.
The survey collected stakeholder demographics and their level of interest (rated on a 5-point scale) in 31 proposed clinical questions, plus additional open-ended responses.
Descriptive statistics and thematic analysis used to analyse the survey data.
Respondents (n = 103) expressed a high level of interest in all proposed questions. Level of positive interest in the 31 proposed clinical questions ranged from 76.70% to 97.96%.
The WCET™ IOG Panel used the data to refine the clinical questions, and to evaluate the importance of topics when completing the adapted evidence-to-decision framework.
Over 40 clinical questions were finalised for consideration.
Incorporating the evidence
The WCET™ IOG Panel used formal processes to identify evidence, complete critical appraisal and data extraction, consider and incorporate evidence.
Literature searches using key terms for each of the clinical questions were conducted in six electronic databases.
Pre-defined inclusion and exclusion criteria used to screen the evidence.
Studies assigned a level of evidence on hierarchy scales for effectiveness and meaningfulness developed by the Joanna Briggs Institute (JBI).
Data extraction tables for each clinical question.
Use of an adapted evidence-to-decision framework to consider the values of stakeholders, benefits, risks, balance of effects, resource requirements, feasibility and acceptability for interventions identified in the research.
Making recommendations and good practice statements based on the body of evidence.
Formal consensus process for good practice statements and expert review to confirm final recommendations.
Your questions, answered
-
The World Council of Enterostomal Therapists® (WCET™) was founded in 1978 to provide an international forum for enterostomal therapy (ET) nurses and a global vehicle for discussion, communication, research, advice and support in the field of ET nursing. The WCET™ is focused on leading the global advancement of specialised professional ostomy, wound and continence nursing care. You can find our more at the WCET™ website.
-
Collaborating organisations were the International Society of University Colon and Rectal Surgeons (ISUCRS) and the International Ostomy Association
-
The full edition will be available in May 2026. The WCET website will have more information when the full IOG (Third edition) is available.
-
A stoma is an opening, either natural or surgically created, which connects a portion of the body cavity to the outside environment. Surgical procedures in which stomata are created end with the suffix -ostomy and begin with a prefix denoting the organ or area in which the stoma is formed.
Colostomy: intestinal stoma formed at the large bowel (colon)
Ileostomy: intestinal stoma formed at the small bowel (ileum)
Urostomy: urinary stoma diverting urine from the bladder, usually done via an ileal conduit from the ureters
-
The recommendations in the WCET™ IOG (Third edition) are focused on people living with an abdominal (intestinal and urinary) ostomy/ostomies or who are preparing for surgery that might lead to formation of a stoma.
The IOG (Third edition) does not address babies, children or teens with a stoma. We suggest reviewing this other guidelines specific to this special interest population. The Global Paediatric Stoma Nurses Advisory Board (2019) and Nurses Specialized in Wound, Ostomy and Continence Canada (in press) have paediatric stoma guidelines.
-
The WCET™ IOG Panel was the governance panel for the WCET™ IOG (Third edition). The Panel consisted of ET nurses from WCET™, colorectal surgeons from ISUCRS, individuals with live experience of stomas and a guideline methodologist. Members of the WCET™ IOG Panel development team are acknowledged here.
The WCET™ IOG Panel invited and coordinated participation, contribution and stakeholder review from:
a Consensus Panel of ET experts
members of the International Ostomy Association
-
Development of the WCET™ IOG (Third edition) was supported by an unrestricted education grant from Hollister Inc.
Evaluation of the evidence

Level of evidence
A strength of recommendation, used for evidence-based recommendations, reflects the trust one can have that implementing the recommendation will lead to more good than harm for most individuals living with a stoma. The strength of recommendation considers the strength of the evidence, as well as the value individuals place on the outcomes, feasibility, acceptability, health equity and resources. We used the GRADE strength of recommendations.
Good practice statements reflect important, accepted and safe clinical practice based on a formal consensus process involving stomal therapy nurse/clinical experts, colorectal surgeons and individual’s with lived experience of a stoma.
Strength of evidence
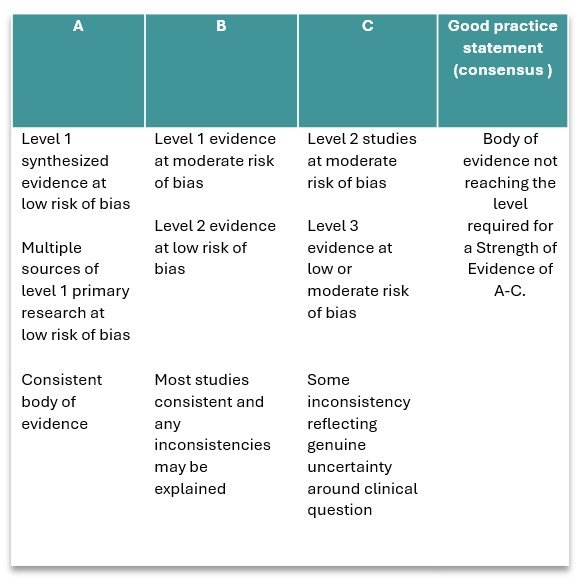
A strength of evidence is the degree of confidence you can have in the research results. It is based on the level of evidence, as well as its risk of bias determined through a formal critical appraisal. The strength of evidence also considers the volume and consistency of the body of evidence. We adapted the National Health and Medical Research Council’s body of evidence matrix.
Stronger evidence comes from well-designed, higher level studies with low risk of bias and with consistent findings across studies. Weaker evidence comes from studies of lower levels of evidence, or at higher risk of bias, or where there is more variation in the findings across studies.
Stronger evidence provides greater confidence in clinical recommendations. Weaker evidence indicates a need for more cautious clinical judgement and shared decision-making.
Strength of recommendation
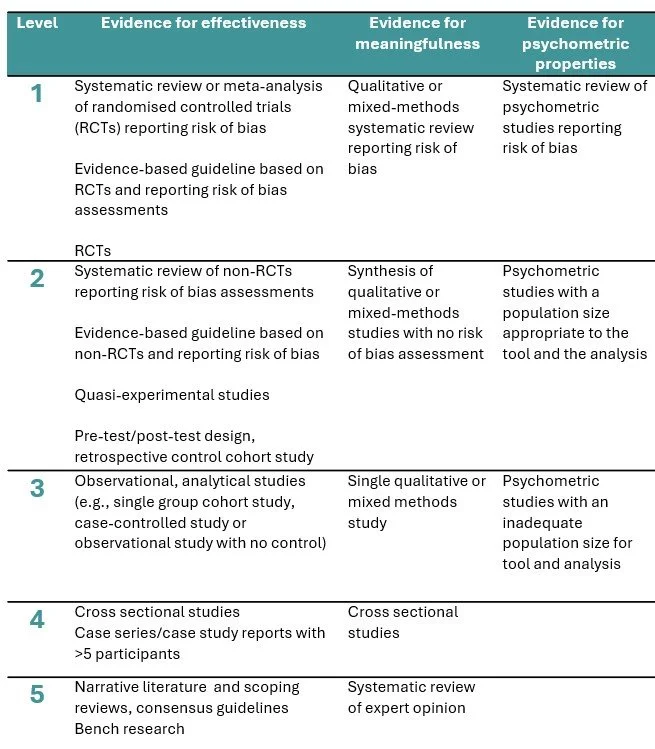
A level of evidence refers to where a study sits on a hierarchical system used to rank research studies according to the methodological quality and validity.
An evidence hierarchy provides a structured way to judge how much confidence can be placed in research findings when informing clinical practice, policy, or guidelines. We used JBI evidence hierarchies.
Higher levels of evidence reflect research designs that minimise systematic error (i.e. higher internal and external validity) and lower levels of evidence are useful for hypothesis generation or informing individualised care in the absence of experimental research.

